
Brain atrophy, commonly referred to as cerebral atrophy, represents a subtle yet progressive neurodegenerative condition marked by the gradual diminution of neurons, the erosion of synaptic connections, and an overall reduction in brain tissue volume. This process can culminate in a wide array of cognitive, emotional, behavioral, and functional impairments that significantly diminish quality of life (Cleveland Clinic, n.d.). Far from being an isolated neurological event, brain atrophy is intricately linked to multiple organ systems, environmental toxins encountered in professional and domestic settings, and broader social dynamics that amplify vulnerability.
Although frequently associated with natural aging processes, neurodegenerative disorders such as Alzheimer's disease, or external factors like traumatic brain injuries, strokes, or chronic alcohol abuse, brain atrophy can also arise from seemingly benign yet modifiable elements. These include persistent nutritional deficiencies, particularly in essential vitamins like B12 (cobalamin), folate (vitamin B9), and B6 (pyridoxine), which collectively contribute to the accumulation of homocysteine—a potent neurotoxic amino acid that instigates oxidative stress, chronic inflammation, and programmed cell death (apoptosis) within neural tissues (Smith et al., 2010). Elevated homocysteine levels, often precipitated by suboptimal B-vitamin intake or absorption, expedite volumetric shrinkage in critical brain regions, including the hippocampus (vital for memory consolidation) and the prefrontal cortex (essential for executive functions like decision-making and impulse control). This biochemical cascade can initiate mild cognitive impairment (MCI), a transitional state that, if left unaddressed, may inexorably progress to full-blown dementia or other cognitive disorders (Vogiatzoglou et al., 2008).
In conventional medical practice, the diagnosis of brain atrophy typically relies on sophisticated neuroimaging techniques, such as magnetic resonance imaging (MRI) or computed tomography (CT) scans, which are often only employed once symptoms have escalated to overt manifestations—such as profound memory deficits, disorientation, linguistic difficulties, or noticeable motor impairments. Early, subtler indicators are frequently overlooked or misattributed to innocuous causes like normal age-related cognitive decline, transient stress, hormonal fluctuations, or even psychological factors such as mild depression (Baptist Health, n.d.). This reactive approach contrasts sharply with the proactive, holistic framework of orthomolecular medicine, a discipline pioneered by two-time Nobel laureate Linus Pauling in the 1960s. Orthomolecular medicine posits that optimal health is achieved by maintaining precise biochemical balances through the administration of nutrients in therapeutic doses, thereby preventing or reversing disease states at their molecular roots (International Society for Orthomolecular Medicine, n.d.).
Practitioners in this field routinely uncover these elusive early symptoms through advanced diagnostic testing, encompassing measurements of active B12 forms (like holotranscobalamin), homocysteine concentrations (where levels exceeding 10 µmol/L are flagged as risky), methylmalonic acid (MMA) as a sensitive marker of B12 functionality, and comprehensive nutrient profiling panels. They contend that standard laboratory reference ranges—such as serum B12 above 200 pg/mL being deemed "normal"—are woefully inadequate for sustaining peak neurological performance and may inadvertently mask the insidious onset of atrophy (Lachner et al., 2012). Moreover, orthomolecular perspectives extend beyond mere nutrition to encompass the interplay of interconnected bodily systems, the insidious infiltration of environmental toxins from occupational and household sources, and the psychosocial stressors that modulate susceptibility, providing a multifaceted lens for safeguarding brain integrity.
The fundamental schism between conventional and orthomolecular paradigms originates from divergent philosophical underpinnings: mainstream medicine adheres rigidly to population-based, evidence-derived diagnostic thresholds and pharmaceutical interventions, often addressing symptoms rather than etiologies. In contrast, orthomolecular medicine champions individualized biochemical optimization, transcending these thresholds to preempt disease progression (International Society for Orthomolecular Medicine, n.d.). Illustratively, borderline low B12 levels ranging from 200 to 400 pg/mL seldom prompt intervention in routine clinical evaluations, yet epidemiological data from large-scale cohort studies reveal their association with accelerated brain volume attrition, approximating 0.5% to 1% per annum in elderly populations (Vogiatzoglou et al., 2008). Compounding this are genetic polymorphisms, such as those in the methylenetetrahydrofolate reductase (MTHFR) gene, which impede efficient folate metabolism and methylation pathways, thereby heightening homocysteine levels and neuronal vulnerability even in the presence of ostensibly adequate dietary nutrient intake (Lachner et al., 2012). Orthomolecular therapeutic regimens seek to elevate B12 concentrations to supramormal levels—typically exceeding 500 pg/mL—employing bioavailable forms like methylcobalamin or hydroxocobalamin, administered orally, sublingually, or via intramuscular injections. This strategy not only mitigates homocysteine toxicity but also bolsters neurogenesis, enhances myelin sheath regeneration, and fortifies endogenous antioxidant systems, potentially halting or even reversing early atrophic changes.
Interconnected Organs: The Systemic Web Underlying Brain Atrophy and B12 Deficiency
Understanding brain atrophy necessitates appreciating its systemic nature, as the brain does not function in vacuo but is enmeshed in a dynamic network of interdependent organs and physiological pathways. The gut-brain axis exemplifies this interconnectivity, serving as a bidirectional communication conduit mediated by neural, hormonal, and immunological signals. Vitamin B12 absorption predominantly transpires in the terminal ileum of the small intestine, contingent upon the binding of intrinsic factor—a glycoprotein secreted by gastric parietal cells in the stomach (Scalabrino, 2009). Autoimmune disorders like pernicious anemia target these parietal cells, ablating intrinsic factor production and precipitating profound B12 malabsorption. This deficiency cascades beyond the gut, engendering neurological sequelae such as subacute combined degeneration of the spinal cord, characterized by demyelination of posterior and lateral columns, resulting in sensory ataxia, paresthesias, and proprioceptive deficits (Reynolds, 2006). Furthermore, intestinal dysbiosis—imbalances in the gut microbiome induced by poor dietary habits, antibiotic overuse, or inflammatory bowel diseases—can further sabotage B12 bioavailability, indirectly fueling hippocampal atrophy and associated memory impairments through disrupted neurotransmitter synthesis and heightened systemic inflammation (Obeid et al., 2007).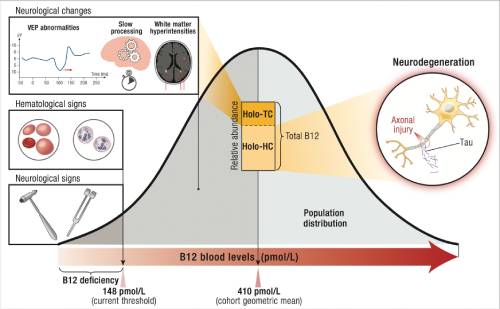
The liver emerges as another pivotal player, functioning as a central hub for homocysteine detoxification via transmethylation and transsulfuration pathways, while also serving as a primary reservoir for B12 storage. Chronic hepatic overload from toxins or metabolic disorders impairs these processes, permitting homocysteine to permeate the blood-brain barrier and incite neuroinflammation, endothelial dysfunction, and accelerated neuronal loss in vulnerable cortical areas (Smith et al., 2010). Renal involvement is equally critical; the kidneys facilitate B12 reabsorption in the proximal tubules, and any compromise—such as in chronic kidney disease, diabetes mellitus, or hypertensive nephropathy—diminishes circulating B12 levels, exacerbating cerebral hypoxia and volumetric decline (American Academy of Neurology, 2011). The hematopoietic system, encompassing bone marrow and blood, is intrinsically linked, as B12 deficiency induces megaloblastic anemia, characterized by enlarged, immature red blood cells that impair oxygen transport to cerebral tissues, thereby potentiating ischemic damage and atrophic progression.
Visual pathways underscore the eye-brain nexus, with B12 scarcity provoking optic atrophy—degeneration of the optic nerve fibers—manifesting as blurred vision, color desaturation, or central scotomas, often preceding broader neurological symptoms (National Health Service, n.d.). Peripheral neuropathy, involving sensory and motor nerves, connects to spinal cord pathology, presenting as glove-and-stocking paresthesias or weakness. In pediatric contexts, maternal B12 deficiency during gestation or lactation can imprint enduring neurodevelopmental deficits on the offspring's central nervous system, affecting cortical maturation and synaptic pruning (Taskesen et al., 2014). Orthomolecular interventions holistically target this web, incorporating B12 supplementation with adjuncts like probiotics for gut microbiota restoration, milk thistle for hepatic support, and renal-protective antioxidants such as coenzyme Q10, thereby addressing multi-organ dysfunction to preserve brain health.
Expanding on cardiovascular interconnections, B12 deficiency and homocysteine elevation contribute to endothelial dysfunction and atherosclerosis, reducing cerebral blood flow and fostering microvascular damage that accelerates atrophy in watershed brain regions. The endocrine system, particularly thyroid function, intersects as hypothyroidism can mimic or coexist with B12 deficiency, amplifying fatigue and cognitive fog through shared metabolic pathways. Musculoskeletal links emerge via neuropathy-induced gait instability, increasing fall risk and secondary brain trauma. Respiratory organs influence via chronic obstructive pulmonary disease (COPD) or sleep apnea, which induce intermittent hypoxia, synergizing with B12 deficits to heighten oxidative neuronal stress. Immune system dysregulation, as in autoimmune gastritis, not only causes B12 malabsorption but also promotes neuroinflammation through cytokine storms. Reproductive organs in women highlight how pregnancy demands escalate B12 needs, with deficiencies risking fetal neural tube defects that persist postnatally as atrophic vulnerabilities. This systemic perspective underscores why orthomolecular assessments often include full-body scans and multi-organ function tests, ensuring comprehensive remediation.
Subtle Symptoms of Brain Atrophy: A Detailed Examination of Cognitive, Mood, Physical, and Behavioral Indicators
The insidious onset of brain atrophy, particularly when rooted in B12 deficiency, often manifests through a constellation of subtle symptoms that evade conventional detection but are readily identified in orthomolecular evaluations. Cognitive domains are frequently the first to exhibit changes, with hippocampal and prefrontal involvement leading to mild memory lapses—such as recurrent forgetfulness of recent conversations, appointments, or where one placed everyday items. These lapses, while superficially resembling benign absentmindedness, tend to worsen under cognitive or emotional stress, reflecting impaired synaptic plasticity and long-term potentiation due to B12-mediated myelin compromise (American Academy of Neurology, 2011). Slowed cognitive processing speed emerges as another hallmark, where individuals experience perceptible delays in assimilating information, solving simple problems, or shifting attention between tasks, akin to the lag in white matter tracts observed in diffusion tensor imaging studies—yet seldom pursued in early stages by standard practitioners (University of California, San Francisco, 2025). Reduced concentration and focus manifest as frequent mind-wandering during reading, conversations, or work, stemming from executive function deficits exacerbated by homocysteine-induced oxidative damage, which orthomolecular therapies counter with B6 and folate synergies to restore dopaminergic balance (Smith et al., 2010). Mild anomia, or word-finding difficulties, involves transient hesitations in retrieving vocabulary during speech, indicative of temporal lobe semantic memory erosion, particularly prevalent in non-hematological B12 deficiencies (Chatterjee et al., 1996). Examples include struggling to name familiar objects or people, often dismissed as "senior moments" but signaling deeper structural changes.
Mood and emotional symptoms add another layer, with subtle irritability or emotional lability arising from amygdala hyperactivity amid nutrient-depleted neurotransmitter environments, leading to disproportionate reactions to minor irritants (National Health Service, n.d.). Subclinical depression presents as persistent apathy, diminished motivation for hobbies, or a vague sense of emotional flatness, linked to serotonin and dopamine dysregulation from impaired methylation (Lachner et al., 2012). Subtle anxiety manifests as low-grade restlessness, rumination on trivial matters, or unexplained worry, reflecting prefrontal cortex and amygdala imbalances aggravated by omega-3 and B-vitamin shortfalls, with orthomolecular remedies like SAMe (S-adenosylmethionine) offering rapid relief.
Physical symptoms include idiopathic fatigue—a pervasive exhaustion unresponsive to rest or caffeine—attributable to mitochondrial dysfunction in energy-intensive neural cells (WebMD, 2024). Intermittent paresthesias, such as tingling or numbness in extremities, herald peripheral neuropathy, while subtle visual disturbances like blurred edges or difficulty with night vision point to optic nerve involvement (Scalabrino, 2009). Sleep architecture disruptions, including delayed onset insomnia or fragmented rest, arise from altered melatonin synthesis and circadian rhythm desynchronization due to B-vitamin deficits (Lone Star Neurology, 2025).
Behavioral and functional alterations encompass reduced mental agility, where adapting to novel situations or planning becomes subtly labored, tied to prefrontal atrophy. Heightened sensitivity to stressors may provoke exaggerated fear or avoidance behaviors, while minor personality shifts—like increased obsessiveness or social withdrawal—signal neurotransmitter imbalances, all potentially reversible with tailored orthomolecular protocols (Lachner et al., 2012). In daily life, these might appear as procrastination on complex tasks, avoidance of social gatherings due to fatigue, or minor errors in judgment, like forgetting bills, which accumulate to erode independence.
Toxins from Worklife: Occupational Exposures as Catalysts for Neurological Deterioration
Occupational environments frequently harbor neurotoxic agents that synergize with B12 deficiencies to precipitate or accelerate brain atrophy, often through mechanisms mimicking nutrient shortfalls. Volatile organic compounds (VOCs), including solvents like toluene, xylene, and benzene found in paints, adhesives, and printing inks, induce chronic solvent encephalopathy—a syndrome of cognitive impairment, mood disorders, and white matter atrophy resembling multiple sclerosis (Occupational Exposure to Solvents, 2015). Workers in manufacturing, auto repair, or dry cleaning may experience subtle executive dysfunction, such as impaired planning or attention, with functional MRI revealing altered brain activation patterns akin to those in B12 deficiency (Occupational Solvent Exposure, 2011). Pesticides, such as organophosphates and carbamates used in agriculture or pest control, inhibit acetylcholinesterase, leading to cholinergic overload and long-term risks for Parkinson's disease, Alzheimer's-like atrophy, and hippocampal volume loss through oxidative stress and mitochondrial disruption (Pesticide-Induced Diseases, n.d.). Farmworkers or landscapers might notice gradual memory decline or tremors, compounded by reduced B12 absorption from gut inflammation.
Heavy metals pose another threat: lead in battery production or plumbing, mercury in dental amalgams or mining, and manganese in welding fumes accumulate in the basal ganglia and cortex, causing optic atrophy, dementia, and parkinsonism by interfering with B12 metabolism and inducing free radical damage (Environmental Toxins and Brain, 2022). Office workers aren't immune; exposure to formaldehyde in building materials or bisphenol A (BPA) in plastics disrupts endocrine function, indirectly elevating homocysteine and fostering prefrontal atrophy. Shift workers face circadian disruption, amplifying B12 utilization and fatigue. Orthomolecular countermeasures include chelation therapy with EDTA for metal removal, alongside high-dose B vitamins and antioxidants like glutathione to neutralize toxins and support detoxification pathways. Real-world examples abound: painters developing "painter's syndrome" with cognitive fog, or welders with manganism exhibiting movement disorders, all underscoring the need for workplace monitoring and nutrient optimization.
Toxic Homelife: Domestic Exposures Silently Eroding Brain Resilience
Domestic settings, ostensibly safe havens, teem with insidious toxins that insidiously undermine brain health, often interfacing with B12 status to hasten atrophy. Quaternary ammonium compounds (quats) in disinfectants and fabric softeners, along with phthalates in air fresheners and vinyl flooring, target oligodendrocytes—the myelin-producing cells—potentially precipitating demyelinating conditions like multiple sclerosis or accelerating atrophy in susceptible individuals (Common Household Chemicals, 2024). Indoor air pollutants from gas stoves, cigarette smoke, or mold growth release fine particulates (PM2.5) and volatile organics that traverse the olfactory pathway or gut-lung-brain axis, inducing neuroinflammation and hippocampal shrinkage (4 Hidden Sources, 2023). Per- and polyfluoroalkyl substances (PFAS), dubbed "forever chemicals" in non-stick cookware, carpets, and waterproof clothing, bioaccumulate and correlate with elevated cholesterol, cognitive decline, and dementia risk by disrupting lipid metabolism and neuronal signaling (Forever Chemicals, 2025).
Legacy contaminants like lead in aging plumbing or paint chips, and methylmercury in contaminated seafood or amalgam fillings, impair neurodevelopment and mimic B12 deficiency symptoms, including neuropathy and visual deficits (Household Chemicals Endanger, 2024). Flame retardants (PBDEs) in furniture and electronics mimic thyroid hormones, altering brain maturation and exacerbating mood instability. Poor ventilation in energy-efficient homes amplifies these effects, with chronic low-level exposure leading to cumulative damage. Orthomolecular strategies advocate home audits, air purifiers, and detoxification protocols using saunas, fiber-rich diets, and supplements like chlorella for binding toxins, coupled with B12 to rebuild neural integrity. Families in older homes might experience collective subtle declines, like children's attention issues or adults' fatigue, highlighting the pervasive threat.
Social Situations: Psychosocial Factors Modulating Atrophy Risk
Social contexts profoundly shape susceptibility to brain atrophy and B12 deficiency, acting as amplifiers of biological vulnerabilities. Socioeconomic inequities restrict access to B12-rich foods like meat, eggs, and dairy, predisposing low-income populations to deficiency and associated MCI (Risk Factors, 2025). Cultural or ethical choices favoring vegetarian or vegan diets without adequate supplementation—common in certain religious or environmental communities—elevate risks, as plant-based sources lack bioavailable B12 (Vogiatzoglou et al., 2008). Chronic psychosocial stress from dysfunctional relationships, workplace bullying, or societal discrimination spikes cortisol, impairing gastric acid production and B12 absorption while accelerating telomeric shortening and hippocampal atrophy (Influences of Vitamin B12, 2022).
Social isolation, prevalent among the elderly or remote workers, fosters depression and reduces dietary variety, compounding B12 depletion and dementia onset (Dementia Associated, 2000). Comorbidities like inflammatory bowel disease or metformin use for diabetes—more common in underserved groups—further deplete B12. Migration or acculturation stresses can disrupt traditional diets, leading to deficiencies. Orthomolecular care incorporates social history assessments, recommending community programs, mindfulness for stress reduction, and group nutrition education alongside supplements. In tight-knit but resource-poor communities, shared deficiencies might manifest as collective cognitive slowdowns, emphasizing social interventions.
Orthomolecular vs. Conventional Approaches: Bridging Philosophical and Practical Divides
Orthomolecular medicine's emphasis on nutrient optimization and toxin mitigation stands in opposition to conventional medicine's symptom-focused, pharmaceutical-centric model, yet integration holds promise (International Society for Orthomolecular Medicine, n.d.). Randomized trials demonstrate B-vitamin supplementation slowing atrophy in MCI patients (Douaud et al., 2013). Conventional limitations include overreliance on hematologic markers, ignoring neurological presentations (Stabler, 2013).
Case Study: A Multifaceted Gradual Descent into Deficiency and Atrophy
The case of a 40-year-old Iranian woman with undiagnosed pernicious anemia illustrates this progression (Abedini et al., 2025). Beginning with subtle oral ulcers and pain in 2014—potentially linked to gastric toxin exposure—symptoms evolved to fatigue, paresthesia, and cognitive fog by 2021, misdiagnosed as fibromyalgia amid social stressors. Critical B12 depletion (<150 pg/mL) was confirmed in 2024, with MRI revealing subcortical lesions suggestive of early atrophy. Intramuscular B12 reversed much, but residual effects lingered.
A parallel infant case from vegetarian maternal diet showed severe cerebral atrophy on MRI, with hypotonia and delays reversed post-supplementation, underscoring intergenerational social influences (Taskesen et al., 2014). Hypothetical scenarios, like a factory worker with solvent exposure developing MCI, highlight occupational-social synergies.
Mechanisms: Biochemical Pathways from Deficiency to Multi-System Damage
At the molecular level, B12 deficiency hampers one-carbon metabolism, stalling DNA synthesis, myelin formation, and neurotransmitter production, leading to demyelination and atrophy (Obeid et al., 2007). Toxins amplify via ROS generation, while social stress activates HPA axis, elevating homocysteine. Conventional diagnostics miss non-anemic cases (Stabler, 2013).
Treatment and Prevention: Comprehensive Strategies for Resilience
Orthomolecular treatments encompass methylcobalamin (1-2 mg daily), folate (5 mg), B6 (50 mg), and antioxidants like vitamin E, monitored via serial labs (International Society for Orthomolecular Medicine, n.d.). Prevention involves toxin avoidance, dietary fortification for at-risk groups, and social support networks (Vogiatzoglou et al., 2008). Lifestyle modifications—exercise, meditation, clean living—bolster efficacy.
In conclusion, the subtle harbingers of brain atrophy demand a panoramic view integrating organs, toxins, and social milieus. Orthomolecular wisdom provides actionable pathways for reversal—empower yourself with knowledge and seek expert guidance.
References
Abedini, M., et al. (2025). A long-standing undiagnosed case of vitamin B12 deficiency. Journal of Medical Case Reports. https://jmedicalcasereports.biomedcentral.com/articles/10.1186/s13256-025-05149-7
American Academy of Neurology. (2011). Low vitamin B12 levels may lead to brain shrinkage, cognitive problems. https://www.aan.com/PressRoom/Home/PressRelease/986
Baptist Health. (n.d.). Cerebral atrophy symptoms & causes. https://www.baptisthealth.com/care-services/conditions-treatments/cerebral-atrophy
Chatterjee, A., et al. (1996). Retardation of myelination due to dietary vitamin B12 deficiency. PubMed. https://pubmed.ncbi.nlm.nih.gov/9028851/
Cleveland Clinic. (n.d.). Vitamin B12 deficiency: Symptoms, causes & treatment. https://my.clevelandclinic.org/health/diseases/22831-vitamin-b12-deficiency
Douaud, G., et al. (2013). Vitamin B12 levels association with functional and structural brain changes. Annals of Neurology. https://onlinelibrary.wiley.com/doi/full/10.1002/ana.27200
International Society for Orthomolecular Medicine. (n.d.). Reversal of Alzheimer's disease and optimization of brain health with orthomolecular medicine. https://isom.ca/article/reversal-alzheimers-disease-optimization-brain-health-orthomolecular-medicine/
Lachner, C., et al. (2012). The neuropsychiatry of vitamin B12 deficiency in elderly patients. The Journal of Neuropsychiatry and Clinical Neurosciences. https://psychiatryonline.org/doi/10.1176/appi.neuropsych.11020052
Lone Star Neurology. (2025). Neurological signs of vitamin B12 deficiency. https://lonestarneurology.net/others/the-neurological-signs-of-vitamin-b12-deficiency-you-shouldnt-ignore/
National Health Service. (n.d.). Vitamin B12 or folate deficiency anaemia - symptoms. https://www.nhs.uk/conditions/vitamin-b12-or-folate-deficiency-anaemia/symptoms/
Obeid, R., et al. (2007). Effects of vitamin B12 and folate deficiency on brain development in children. PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC3137939/
Reynolds, E. (2006). Vitamin B12, folic acid, and the nervous system. The Lancet Neurology. https://www.thelancet.com/journals/laneur/article/PIIS1474-4422(06)70598-9/fulltext
Scalabrino, G. (2009). Cobalamin deficiency: Clinical picture and radiological findings. PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC3847746/
Smith, A. D., et al. (2010). Brain atrophy and B vitamins. Alzheimer's Association. https://www.alz.org/blog/2010/brain-atrophy-and-b-vitamins
Stabler, S. P. (2013). Vitamin B12 and cognitive function: An evidence-based analysis. PubMed. https://pubmed.ncbi.nlm.nih.gov/24379897/
Taskesen, M., et al. (2014). Cerebral atrophy in a vitamin B12-deficient infant of a vegetarian mother. PubMed. https://pubmed.ncbi.nlm.nih.gov/25076673/
University of California, San Francisco. (2025). 'Healthy' vitamin B12 levels not enough to ward off neuro decline. https://www.ucsf.edu/news/2025/02/429491/healthy-vitamin-b12-levels-not-enough-ward-neuro-decline
Vogiatzoglou, A., et al. (2008). Vitamin B12 status and rate of brain volume loss in community-dwelling elderly. PubMed. https://pubmed.ncbi.nlm.nih.gov/18779510/
WebMD. (2024). Pictures: Signs you're low on vitamin B12. https://www.webmd.com/vitamins-and-supplements/ss/slideshow-signs-lack-vitamin-b12
Environmental Toxins and Brain: Life on Earth is in Danger. (2022). PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC9540824/
Pesticide-Induced Diseases: Brain and Nervous System Disorders. (n.d.). Beyond Pesticides. https://www.beyondpesticides.org/resources/pesticide-induced-diseases-database/brain-and-nervous-system-disorders
Occupational Solvent Exposure and Brain Function: An fMRI Study. (2011). PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC3222975/
Occupational Exposure to Solvents: Neuropsychiatric and Imaging Features. (2015). Psychiatry Online. https://psychiatryonline.org/doi/10.1176/appi.neuropsych.270101
Common Household Chemicals Pose New Threat to Brain Health. (2024). Case Western Reserve University. https://case.edu/news/common-household-chemicals-pose-new-threat-brain-health
Effects of Environmental Toxins on Brain Health and Development. (2023). PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC9945219/
Brain Health: 2 Groups of Household Chemicals Linked to Cell Damage. (2024). Medical News Today. https://www.medicalnewstoday.com/articles/household-chemicals-may-increase-risk-neurological-conditions-autism-ms
4 Hidden Sources of Brain Toxins in Your Home. (2023). Psychology Today. https://www.psychologytoday.com/us/blog/the-modern-brain/202310/4-hidden-sources-of-brain-toxins-in-your-home
Forever Chemicals: The Link Between PFAS and Dementia. (2025). University of Miami. https://news.med.miami.edu/forever-chemicals-the-link-between-pfas-and-dementia/
Household Chemicals Endanger Brain's Myelin-Forming Cells. (2024). News Medical. https://www.news-medical.net/news/20240326/Household-chemicals-endanger-brains-myelin-forming-cells.aspx
Influences of Vitamin B12 Supplementation on Cognition and Homocysteine. (2022). PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC9002374/
Risk Factors and Comorbidities Associated With Vitamin B12 Deficiency. (2025). SAGE Journals. https://journals.sagepub.com/doi/10.1177/21501319251360498
Dementia Associated With Vitamin B12 Deficiency. (2000). Psychiatry Online. https://psychiatryonline.org/doi/full/10.1176/jnp.12.3.389
- Hits: 170










